
Ph.D. Professor Kenji Chamoto
Our division aims to achieve an integrated understanding of the regulatory mechanisms of anti-tumor immune responses at the molecular, cellular, and metabolic levels, and to translate these insights into next-generation cancer immunotherapies. We particularly focus on overcoming primary and acquired resistance to immune checkpoint blockade by elucidating metabolic reprogramming and mitochondrial regulation in immune cells. By cross-disciplinary analyses of metabolic stress, chronic inflammation, and aging-associated immune dysfunction within the tumor microenvironment, we promote bidirectional translational research to develop mechanism-based and clinically effective therapeutic strategies.
Research and Education
Immune checkpoint blockade has revolutionized cancer therapy; however, more than half of patients still fail to achieve durable clinical benefit, and both primary resistance and acquired resistance remain major challenges. Our division focuses on elucidating the molecular mechanisms underlying these limitations, particularly from the perspective that the metabolic state of immune cells critically determines the quality, persistence, and therapeutic responsiveness of anti-tumor immunity. We investigate immunometabolic regulatory networks centered on mitochondrial function and examine how metabolic stresses within the tumor microenvironment—including hypoxia, oxidative stress, and inflammatory signaling—affect T-cell differentiation and exhaustion, as well as macrophage-mediated immunosuppression at the molecular and cellular levels. In addition, we analyze how aging-associated immune alterations and chronic inflammatory conditions reshape anti-tumor immune responses. By integrating cancer, aging, and inflammation in a cross-disciplinary framework, we seek to define universal principles governing immune regulation. These insights will guide the identification of novel therapeutic targets, the design of rational combination strategies with existing immunotherapies, and their translation into clinical applications.
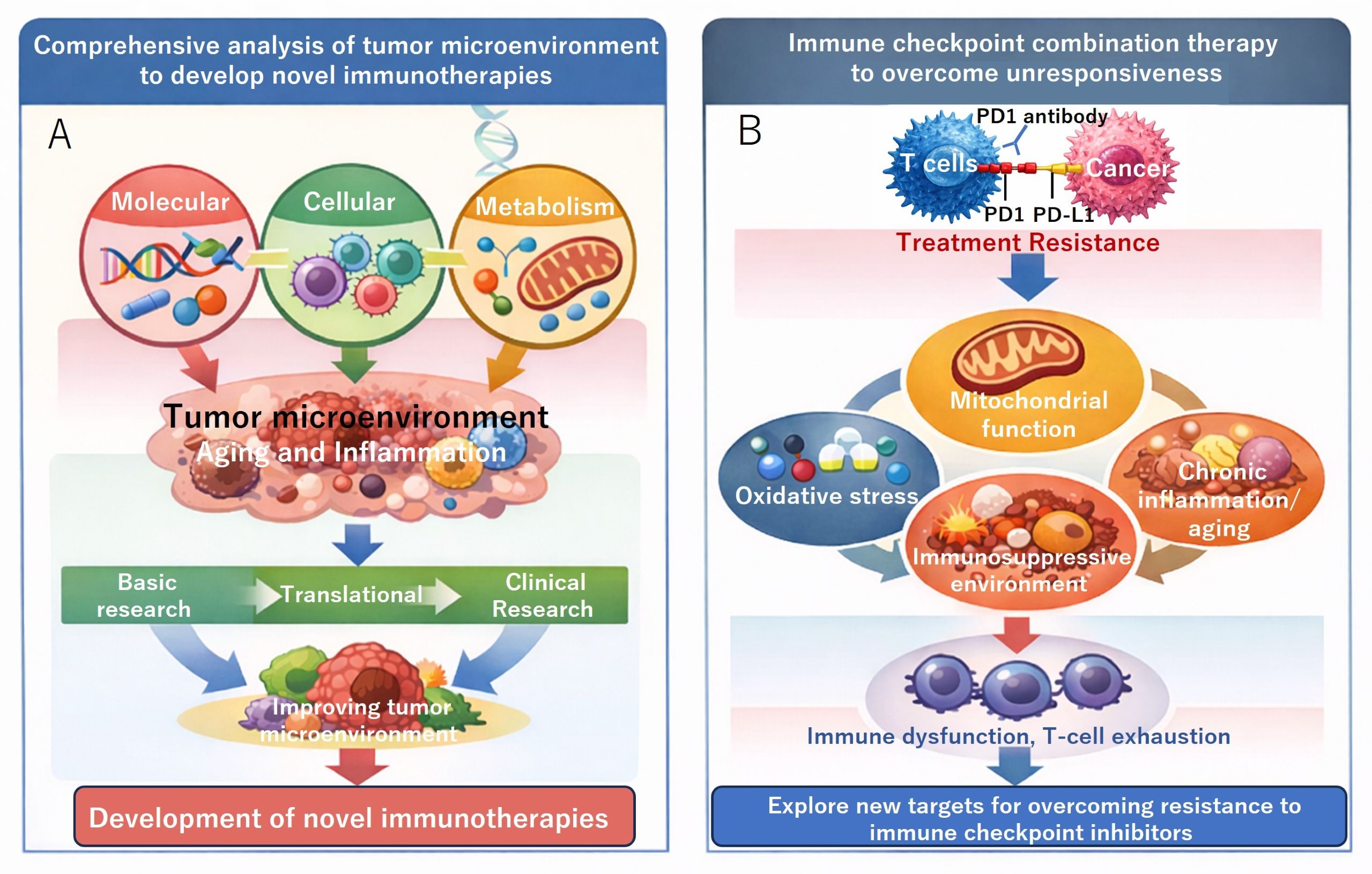 A) We promote translational research (TR) grounded in novel insights derived from an integrated, interdisciplinary understanding of the tumor and inflammatory microenvironment at the cellular, metabolic, and molecular levels. Challenges encountered during the translational process are systematically addressed through reverse translational research, whereby clinical observations are fed back into basic investigation. Through this bidirectional framework, we aim to develop clinically effective immunotherapies based on solid mechanistic foundations.
A) We promote translational research (TR) grounded in novel insights derived from an integrated, interdisciplinary understanding of the tumor and inflammatory microenvironment at the cellular, metabolic, and molecular levels. Challenges encountered during the translational process are systematically addressed through reverse translational research, whereby clinical observations are fed back into basic investigation. Through this bidirectional framework, we aim to develop clinically effective immunotherapies based on solid mechanistic foundations.
B) We investigate the molecular mechanisms underlying resistance to PD-1 blockade from the perspectives of mitochondrial function, oxidative stress, and aging-associated immune alterations. By elucidating how these factors contribute to T-cell dysfunction and exhaustion, we seek to establish rational combination strategies that enhance therapeutic efficacy and overcome resistance to immune checkpoint inhibitors.
Recent Publications
- Haku Y, Kitaoka K, Ichimaru K, Hirano T, Wang J, Sonomura K, Maruo A, Hirose S, Wang Y, Ito K, Kozuki T, Yurimoto K, Kiyono M, Kosako H, Menju T, Date H, Kobayashi T, Omori K, Yaguchi Y, Honjo T and Chamoto K*(*corresponding). Active aldehydes accelerate CD8+ T cell exhaustion by metabolic alteration in the tumor microenvironment. (2026) Nat Immunol. 27(2): 281-294.
- Zhang R, Ma R, Leong MML, Watson IR, Iida K, Yaguchi T, Matsuda F, Honjo T, Chamoto K*(*corresponding). Chimeric MHC class I- and II-restricted non-self epitopes broaden antitumor T cell reactions. (2026) J Exp Med. 223(2):e20250025.
- Chamoto K, Yaguchi T, Tajima M, Honjo T. Insights from 30-year journey: function, regulation and therapeutic modulation of PD1. (2023) Nat Rev Immnol. 23(10):682-695.
- Tanaka K*, Chamoto K*(*co-corresponding), Saeki S, Hatae R, Ikematsu Y, Sakai K, Ando N, Sonomura K, Kojima S, Taketsuna M, Kim YH, Yoshida H, Ozasa H, Sakamori Y, Hirano T, Matsuda F, Hirai T, Nishio K, Sakagami T, Fukushima M, Nakanishi Y, Honjo T, Okamoto I. Combination bezafibrate and nivolumab treatment of patients with advanced non–small cell lung cancer. (2022) Sci Transl Med. 14 (675): eabq0021.
- Al-Habsi M*, Chamoto K*, Matsumoto K*, Nomura N*(*equally contributed), Zhang B, Sugiura Y, Sonomura K, Maharani A, Nakajima Y, Wu Y, Nomura Y, Menzies R, Tajima M, Kitaoka K, Haku Y, Delghandi S, Yurimoto K, Matsuda F, Iwata S, Ogura T, Fagarasan S, Honjo T. Spermidine activates mitochondrial trifunctional protein and improves antitumor immunity in mice. (2022) Science. 28; 378 (6618): eabj3510.
Laboratory
Professor: Kenji Chamoto
Email: chamoto.kenji.4w@kyoto-u.ac.jp
