Alternative splicing of pre-mRNA generates a highly dynamic human proteome through networks of coordinated splicing events, and is regulated on tissue-specific stage-dependent way. Our goal is to establish a new paradigm on the mechanism of morphogenesis from the view point of mRNA processing regulation. We will also try to find chemical modifiers of mRNA processing, which will be applicable for clinical therapy against malignant tumors, virus infections and neuromuscular diseases caused by aberrant splicing. We welcome enthusiastic young researchers who will challenge to open new fields of biology.
Research and Education
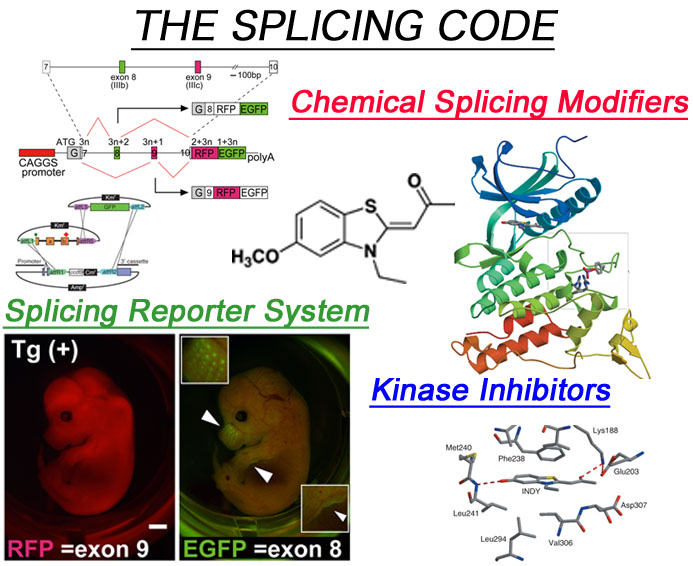
- Decipherment of splicing code
Alternative splicing events are dynamically regulated in developmental stage-dependent and tissue specific manners, and regulated by ‘splicing code’ made up of loosely defined consensus sequences. To decipher the splicing code, we have developed transgenic alternative splicing reporter systems that enable us to visualize alternative splicing events in living cells. - Development of new drugs to cure RNA diseases
There is a growing awareness that misregulations of mRNA processing are causative of many human diseases. We focused on the mRNA splicing process, and have developed compounds that alter the alternative splicing events or even reverse some misregulated splicing events. - Chemical biology
We are one of pioneers of chemical biology field, Chemical biology is one of new and rapidly growing research fields, and we are one of pioneer groups of this field. Using new techniques of chemical biology, we are challenging to develop novel drugs to cure hereditary diseases, cancers, virus infections, and other incurable diseases.
Recent Publications
- Matsushima S, Ajiro M, Iida K, Chamoto K, Honjo T, Hagiwara M. (2022) Chemical induction of splice-neoantigens attenuates tumor growth in a preclinical model of colorectal cancer. Sci Transl Med. 14(673):eabn6056.
- Maeda S, Shiimura Y, Asada H, Hirata K, Luo F, Nagano E, Tanaka N, Toyomoto M, Inoue A, Aoki J, Iwata S, Hagiwara M. (2021) Endogenous agonist-bound S1PR3 structure reveals determinants of G protein-subtype bias. Sci Adv. 7(24):eabf5325.
- Ajiro M, Awaya T, Kim YJ, Iida K, Denawa M, Tanaka N, Kurosawa R, Matsushima S, Shibata S, Sakamoto T, Studer L, Krainer AR, Hagiwara M. (2021) Therapeutic manipulation of IKBKAP mis-splicing with a small molecule to cure familial dysautonomia. Nat Commun. 12(1):4507.
- Toyomoto M, Inoue A, Iida K, Denawa M, Kii I, Ngako Kadji FM, Kishi T, Im D, Shimamura T, Onogi H, Yoshida S, Iawata S, Aoki J, Hosoya T, Hagiwara M. (2021) S1PR3-G12-biased agonist ALESIA targets cancer metabolism and promotes glucose starvation. Cell Chem Biol. S2451-9456(21)00004-0.
- Nakano-Kobayashi A, Fukumoto A, Morizane A, Nguyen DT, Le TM, Hashida K, Hosoya T, Takahashi R, Takahashi J, Hori O, Hagiwara M. (2020) Therapeutics potentiating microglial p21-Nrf2 axis can rescue neurodegeneration caused by neuroinflammation. Sci Adv. 6(46):eabc1428.
