
Ph.D. Professor Mitsuharu Midorikawa
Our body consists of tens of trillions of cells, and exocytosis plays a crucial role in cellular communication. We are focusing on the smallest (~ 40 nm in diameter) and the fastest (within 1 ms) exocytosis in our body, i.e., exocytosis of the synaptic vesicles. We aim to elucidate how this slight biological reaction varies in different brain regions or under different circumstances to generate different properties of neural circuits.
Research and Education
We mainly focus on the exocytosis mechanism of the neuron in the mouse central nervous system. To precisely measure the synaptic vesicles, of which diameter is only ~ 40 nm, to fuse with the presynaptic plasma membrane, we mainly use electrophysiology and live imaging technique. In addition, we are incorporating techniques such as super-resolution microscopy to visualize the molecular organization at the nanometer level and morphological methods to visualize the wiring of neural circuits on a single-fiber level.
By using these techniques to investigate differences between different neurons, as well as plastic changes resulting from development, adaptation, and learning, we aim to unravel how variations in exocytosis properties affect neural circuits and even higher brain functions. We aim to bridge our study to creating therapeutic strategies for developmental and mental disorders, neurorehabilitation, and beyond.
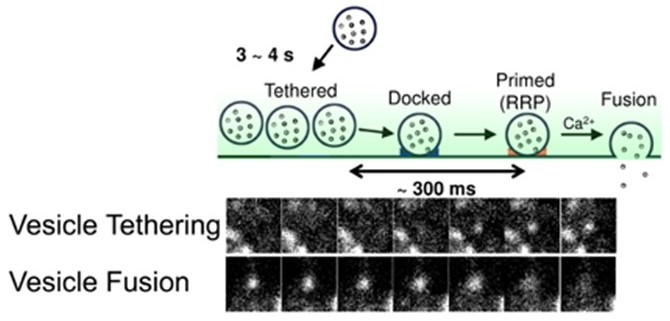 The tethering of the single synaptic vesicle to the plasma membrane (upper) and fusion (lower) at the mouse CNS neuron.
The tethering of the single synaptic vesicle to the plasma membrane (upper) and fusion (lower) at the mouse CNS neuron.
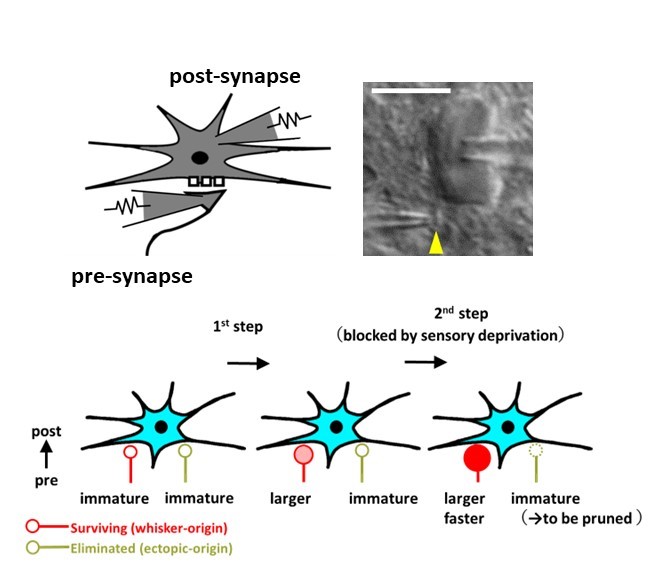 Direct electrophysiological recordings from presynaptic terminals and postsynaptic cells (upper) reveal the time course of presynaptic functional development at the mouse somatosensory thalamus (lower).
Direct electrophysiological recordings from presynaptic terminals and postsynaptic cells (upper) reveal the time course of presynaptic functional development at the mouse somatosensory thalamus (lower).
Recent Publications:
- Midorikawa M, Miyata M. (2021). Distinct functional developments of surviving and eliminated presynaptic terminals. Proceedings of the National Academy of Sciences of the United States of America, 118(11): e2022423118.
- Midorikawa M, Sakaba T. (2017). Kinetics of Releasable Synaptic Vesicles and Their Plastic Changes at Hippocampal Mossy Fiber Synapses. Neuron, 96(5), 1033-1040.
- Okamoto Y, Lipstein N, Hua Y, Lin KH, Brose N, Sakaba T, Midorikawa M. (2016). Distinct modes of endocytotic presynaptic membrane and protein uptake at the calyx of Held terminal of rats and mice. eLife, 5:e14643.
- Midorikawa M, Sakaba T. (2015). Imaging Exocytosis of Single Synaptic Vesicles at a Fast CNS Presynaptic Terminal. Neuron, 88(3), 492-8.
- Midorikawa M, Tsukamoto Y, Berglund K, Ishii M, Tachibana M. (2007). Different roles of ribbon-associated and ribbon-free active zones in retinal bipolar cells. Nature Neuroscience, 10(10), 1268-76.
Laboratory
[Professor] Mitsuharu Midorikawa, Ph.D.
TEL:+81-75-751-3913
e-mail:midorikawa.mitsuharu.3y atmark kyoto-u.ac.jp
(please change atmark to @)
