
M.D., Ph.D. Professor Akiyuki Taruno
Regardless of conscious awareness, the human body maintains homeostasis by sensing internal and external environmental changes and responding appropriately. Disruption of these processes leads to diverse diseases. However, many aspects of sensory function remain unexplained, and previously unidentified sensory cells may exist. In addition, how information transmitted from diverse sensory organs to the brain is integrated by neural circuits and translated into appropriate responses remains largely unknown. Through studies in mammals, we aim to elucidate new principles of sensory systems and to advance their medical and physiological applications.
Research and Education
Humans eat approximately 90,000 times over the course of a lifetime. Feeding/drinking behavior is regulated in a complex and highly precise manner in response to changes in internal and external environments, yet many fundamental questions remain regarding the sensory mechanisms underlying information detection and integration. Disruption of these processes contributes to lifestyle-related diseases, including hypertension.
Our work has revealed that taste signal transmission from taste buds to gustatory nerves via channel synapses, sodium taste reception, and the molecular and cellular mechanisms controlling swallowing and cough reflexes differ markedly from conventional concepts. These findings suggest that sensory reception and transmission mechanisms have evolved in diverse ways depending on stimulus modality.
With the goal of elucidating sensory networks that govern physiological homeostasis, we investigate molecular, cellular, and neural circuit mechanisms underlying gustatory and visceral sensory processing and plasticity; central mechanisms linking multisensory integration to behavioral and physiological outputs; atypical and previously uncharacterized sensory systems; disease mechanisms in sensory pathways and their therapeutic manipulation; and the biological functions of channel synapses.
In education, we aim to cultivate individuals who pursue original research approaches, remain resilient to prevailing trends and adversity, and thrive internationally.
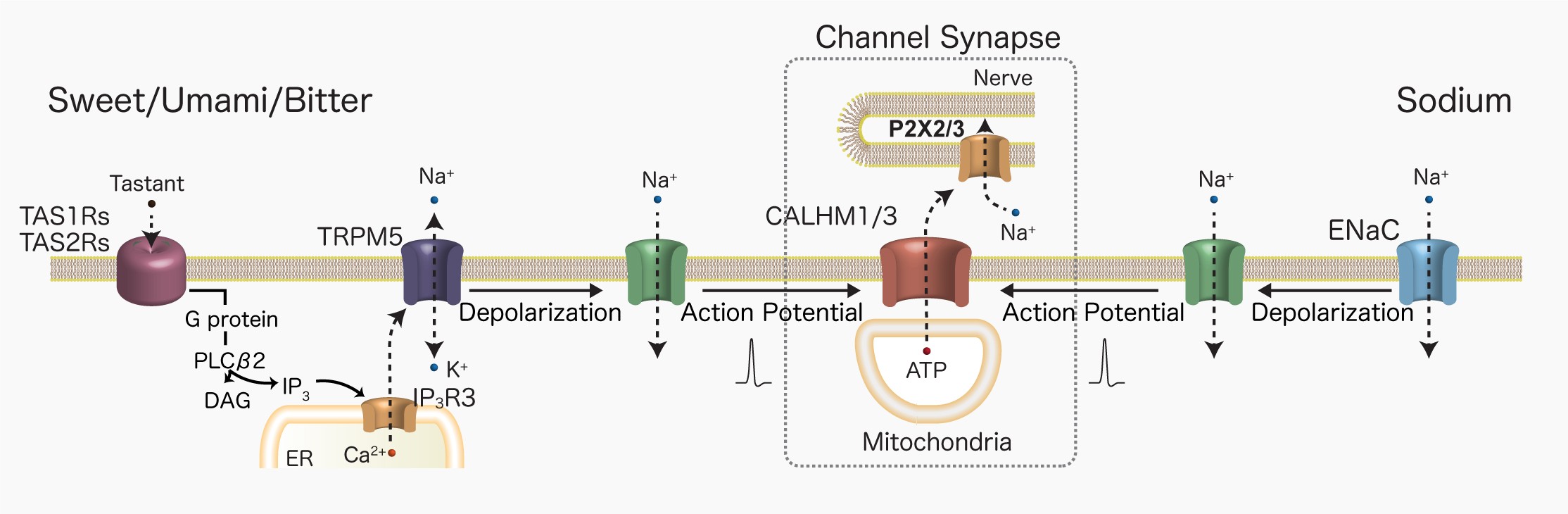 – Channel Synapses and Molecular Mechanisms of Taste Reception
– Channel Synapses and Molecular Mechanisms of Taste Reception
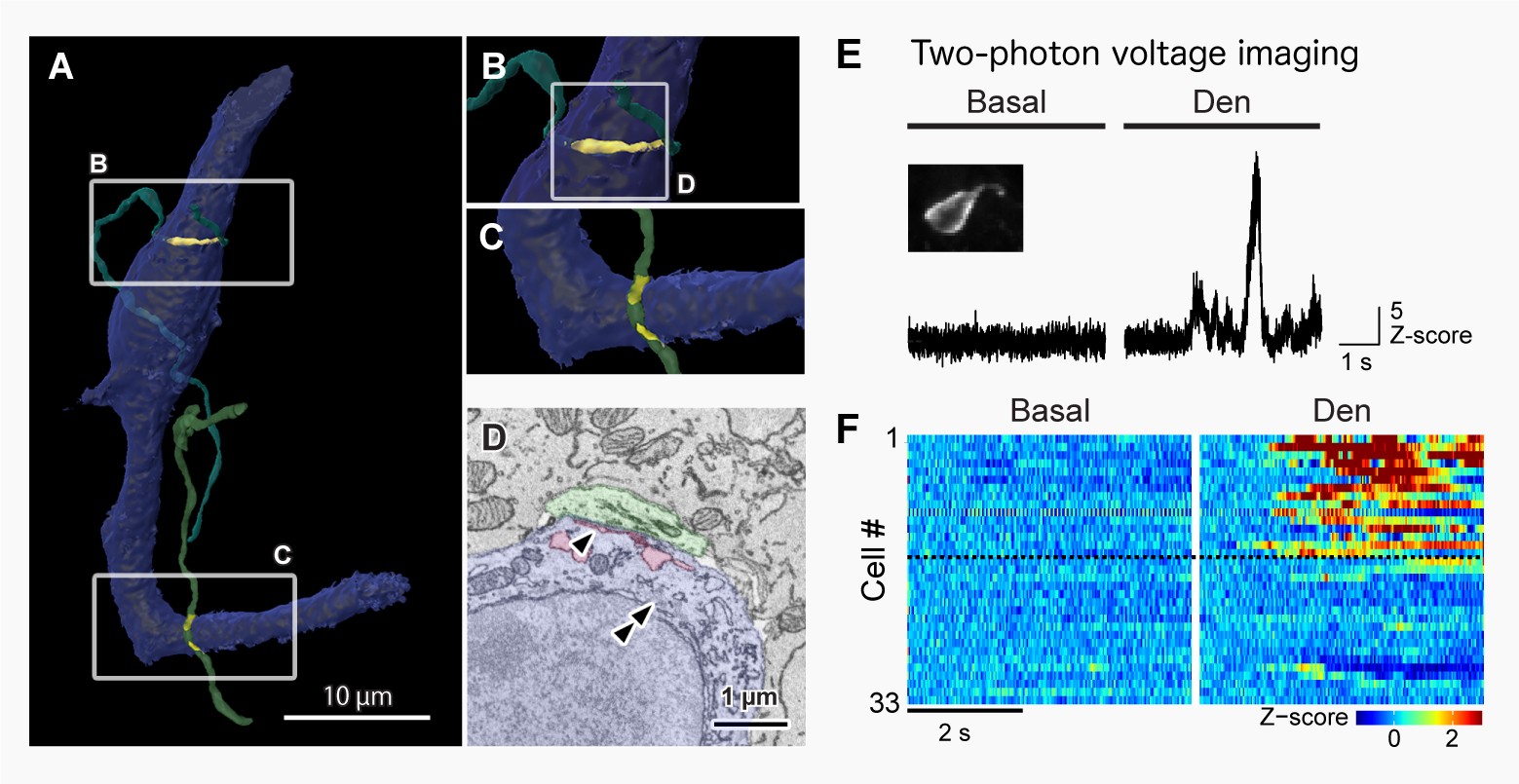 – Rare Sensory Epithelial Cells Generating Action Potentials
– Rare Sensory Epithelial Cells Generating Action Potentials
(A–D) Three-dimensional reconstruction images (A–C) and synaptic structure (D) of laryngeal tuft cells captured using 3D-CLEM.
Blue: tuft cells; Green: vagal nerve.
Arrowheads: endoplasmic reticulum; double arrowheads: filamentous structures.
(E–F) Action potentials generated in tuft cells in response to chemical stimulation (Den), visualized by membrane potential imaging.
Recent Publications
- Soma S, Hayatsu N, Nomura K, Sherwood MW, Murakami T, Sugiyama Y, Suematsu N, Aoki T, Yamada Y, Asayama M, Kaneko M, Ohbayashi K, Arizono M, Ohtsuka M, Hamada S, Matsumoto I, Iwasaki Y,Ohno N, Okazaki Y, Taruno A. Channel synapse mediates neurotransmission of airway protective chemoreflexes. Cell 188; 2687-2704, 2025.
- Demura K, Kusakizako T, Shihoya W, Hiraizumi M, Nomura K, Shimada H, Yamashita K, Nishizawa T, Taruno A, Nureki O. Cryo-EM structures of calcium homeostasis modulator channels in diverse oligomeric assemblies. Sci Adv 6; eaba8105, 2020.
- Nomura K, Nakanishi M, Ishidate F, Iwata K, Taruno A. All-electrical Ca2+-independent transduction mediates attractive sodium taste in taste buds. Neuron 106; 816-829, 2020.
- Ma Z, Taruno A, Ohmoto M, Jyotaki M, Lim JC, Miyazaki H, Niisato N, Marunaka Y, Lee RJ, Hoff H, Payne R, Demuro A, Parker l, Mitchell C, Henao-Mejia J, Tanis JE, Matsumoto I, Tordoff MG, Foskett JK. CALHM3 is essential for rapid ion channel-mediated purinergic neurotransmission of GPCR-mediated tastes. Neuron 98; 547-561, 2018.
- Taruno A, Vingtdeux V, Ohmoto M, Ma Z, Dvoryanchikov G, Li A, Adrien L, Zhao H, Leung S, Abernethy M, Koppel J, Davies P, Civan M M, Chaudhari N, Matsumoto I, Hellekant G, Tordoff MG, Marambaud P, Foskett JK. CALHM1 ion channel mediates purinergic neurotransmission of sweet, bitter and umami tastes. Nature 495; 223-226, 2013.
Laboratory
Professor: Akiyuki Taruno, M.D. & Ph.D.
Assistant Professor: Kengo Nomura, Ph.D.
TEL:not determined
Email:taruno.akiyuki.7a(at)kyoto-u.ac.jp
URL:https://cmp.med.kyoto-u.ac.jp
