
M.D., Ph.D. Professor Shimpei Gotoh
The lung is a vital organ for gas exchange throughout the entire body and has a well-developed function to exclude foreign matters and pathogens due to its direct contact with the air space. The two major regions, airways and alveoli, harbor tissue stem cells contributing to repair of the lung after a variety of injuries. Our laboratory is highly interested in intractable lung diseases and working on clarifying their pathomechanisms for finding biomarkers and therapeutic agents as well as developing new methods for lung regeneration.
Research and Education
We have pioneered the new field of lung regenerative medicine, starting with the establishment of the methods to generate airway and alveolar cells from human pluripotent stem cells. We are studying various intractable lung diseases, including rare diseases, using two major approaches: one is to establish disease models to search for biomarkers and develop therapeutic agents; the other is to develop a method of regenerating/reconstructing lungs using cells. Lung transplantation has been an established therapeutic option for patients with irreversible respiratory failure, but the shortage of donors is a serious issue on a global level. We sincerely hope to contribute to therapeutic options to regenerate lungs in the future. In the graduate school, students conduct research activities at an international level to be a global researcher who can lead the future science community and medicine, while participating in events at the Center for iPS Cell Research and Application (CiRA) and Graduate School of Medicine, Kyoto University. We expect applicants who wish to concentrate on respiratory research with their passion.
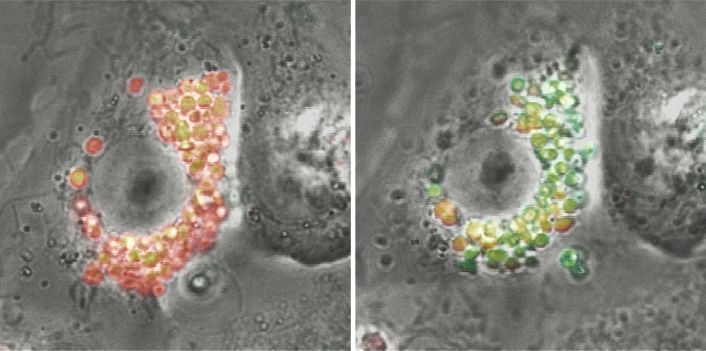 Fig.1. Live cell imaging of human iPS cell-derived alveolar type II cells. Release of pulmonary surfactant upon stimulation was visualized. Red: lamellar bodies, Green: released phosphatidylcholine.
Fig.1. Live cell imaging of human iPS cell-derived alveolar type II cells. Release of pulmonary surfactant upon stimulation was visualized. Red: lamellar bodies, Green: released phosphatidylcholine.
Korogi Y et al. In vitro disease modeling of Hermansky-Pudlak syndrome type 2 using human induced pluripotent stem cell-derived alveolar organoids. Stem Cell Reports. 2019;12:431-440.
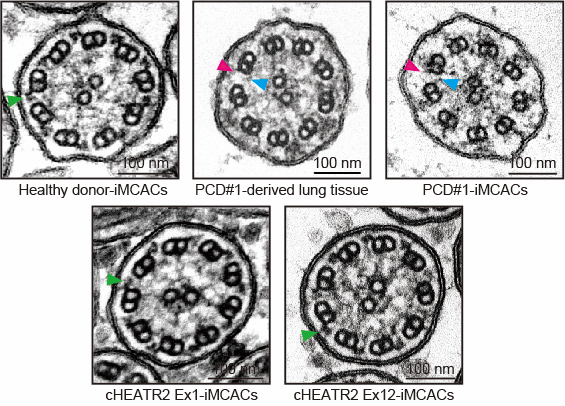 Airway epithelial cells derived from human iPS cells generated from a patient with primary ciliary dyskinesia (PCD) were used to recapitulate the pathophysiology of the disease in vitro. (The photo shows electron microscopic images of multiciliated airway cells. The upper left figure is derived from a healthy donor, the upper middle figure from the patient lung tissue, the upper right figure from the patient iPS cells, and the lower figure from repaired iPS cells of each allele of the PCD causative gene. Lack of outer dynein arms (ODA) and inner dynein arms (IDA) indicated by magenta and blue arrowheads, respectively. Green arrowhead indicates the ODA.)
Airway epithelial cells derived from human iPS cells generated from a patient with primary ciliary dyskinesia (PCD) were used to recapitulate the pathophysiology of the disease in vitro. (The photo shows electron microscopic images of multiciliated airway cells. The upper left figure is derived from a healthy donor, the upper middle figure from the patient lung tissue, the upper right figure from the patient iPS cells, and the lower figure from repaired iPS cells of each allele of the PCD causative gene. Lack of outer dynein arms (ODA) and inner dynein arms (IDA) indicated by magenta and blue arrowheads, respectively. Green arrowhead indicates the ODA.)
Sone N, et al. Multicellular modeling of ciliopathy by combining iPS cells and microfluidic airway-on-a-chip technology. Sci Transl Med. 2021;13 (601), eabb1298.
Publications
- Ohnishi Y, Masui A, Suezawa T, Mikawa R, Hirai T, Hagiwara M, Gotoh S. Screening of factors inducing alveolar type 1 epithelial cells using human pluripotent stem cells. Stem Cell Rep. 2024; 4(20).
- Masui A, Hashimoto R, Matsumura Y, Yamamoto T, Nagao M, Noda T, Takayama K, Gotoh S. Micro-patterned culture of iPSC-derived alveolar and airway cells distinguishes SARS-CoV-2 variants. Stem Cell Rep. 2024; 4(20).
- Tamai K, Sakai K, Yamaki H, Moriguchi K, Igura K, Maehana S, Suezawa T, Takehara K, Hagiwara M, Hirai T, Gotoh S. iPS cell-derived mesenchymal cells that support alveolar organoid development. Cell Rep Methods. 2022; 2(10):100314.
- Ikeo S, Yamamoto Y, Ikeda K, Sone N, Korogi Y, Tomiyama L, Matsumoto H, Hirai T, Hagiwara M, Gotoh S. Core-shell hydrogel microfiber-expanded pluripotent stem cell-derived lung progenitors applicable to lung reconstruction in vivo. Biomaterials. 2021; 276:121031
- Sone N, Konishi S, Igura K, Tamai K, Ikeo S, Korogi Y, Kanagaki S, Namba T, Yamamoto Y, Xu Y, Takeuchi K, Adachi Y, Chen-Yoshikawa TF, Date H, Hagiwara M, Tsukita S, Hirai T, Torisawa Y, Gotoh S. Multicellular modeling of ciliopathy by combining iPS cells and microfluidic airway-on-a-chip technology. Sci Transl Med. 2021; 13: eabb1298.
Laboratory
M.D., Ph.D. Professor: Shimpei Gotoh
TEL:+81-75-366-7342
FAX:+81-75-366-7341
E-mail:gotoh-g@cira.kyoto-u.ac.jp
