
Ph.D. Program-specific Professor Shiro Tanaka
Health technologies such as drugs should be assessed for efficacy and safety before applied to clinical practice through clinical trials, which require involvement of statisticians who have expertise in biostatistics. Clinical Biostatistics is a discipline on the statistical methodology in clinical trials such as study design and statistical analysis of clinical trial data.
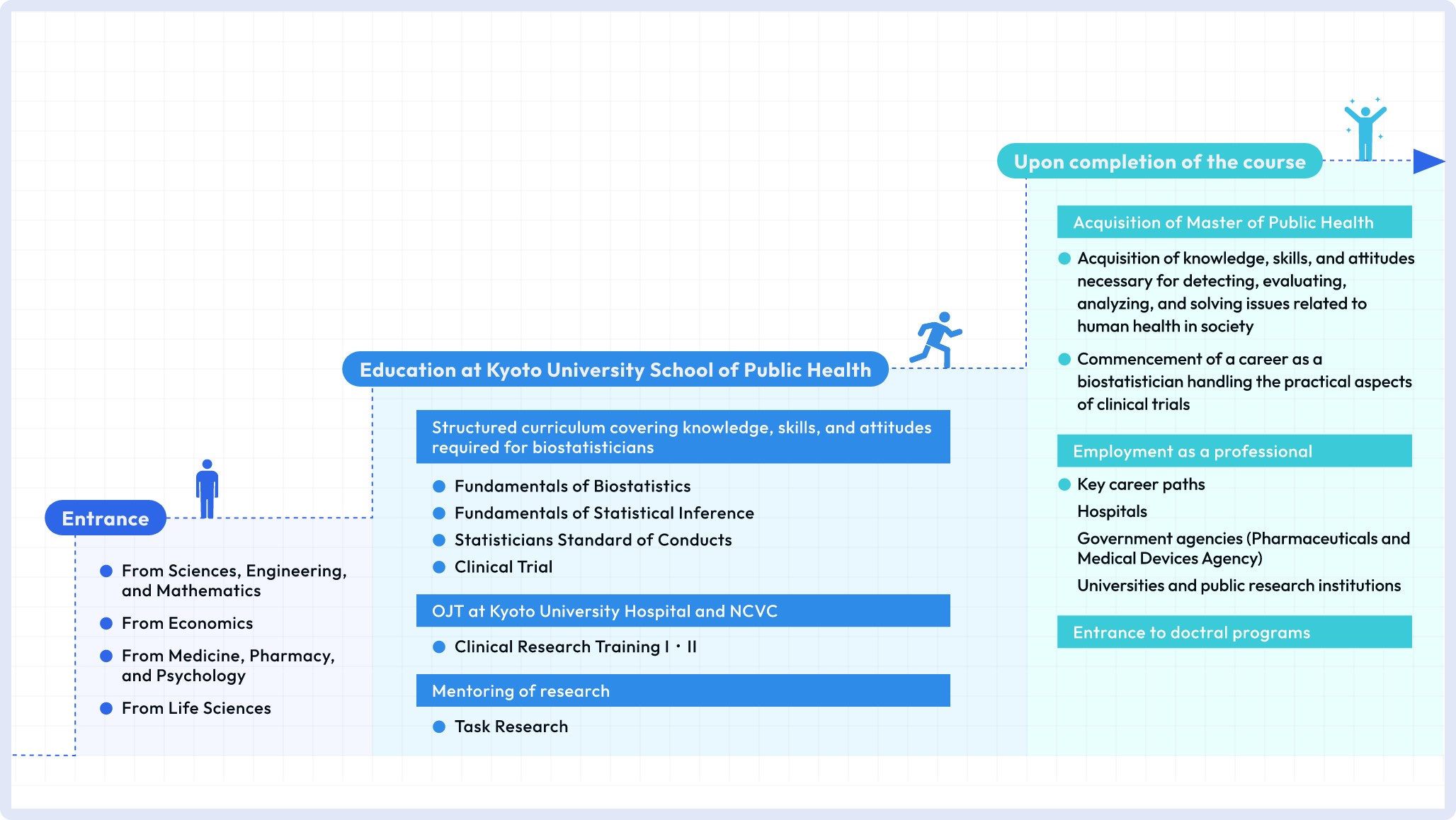
Clinical Biostatistics Course is a course on biostatistics conducted by Department of Clinical Biostatistics and Department of Biostatistics. This course is intended for graduates from any fields, i.e. mathematics, physics, economics, life science and medicine. Medical knowledge is not a prerequisite.
Research and Education
Clinical Biostatistics Course is a two-year Professional Degree Course (=master course) established in Kyoto University School of Public health (SPH). Master of Public Health (MPH [professional degree]) will be awarded in completing the course. This course provides students with, not only mandatory subjects such as basic statistics and clinical biostatistics classes, but also on-the-job training at Kyoto University Hospital and National Cerebral and Cardiovascular Center which gives students opportunities to experience the practice of academic clinical trials. This course is funded by the Japan Agency for Medical Research and Development (AMED).
Research and development
- Develop an educational program for Clinical Biostatistics Course
-
Open attendance courses
- Biostatistics for clinical researchers (seminars at hospitals)
- Introduction to statistics for undergraduate students (E-learning contents for Open Course Ware )
- Statistical analysis of medical and health database
- Statistical methods for missing data in clinical trials
Recent Publications
- Okada K, Tanaka S, Matsubayashi J, Takahashi K, Yokota I. De-coupling power and type I error rate considerations when incorporating historical control data using a test-then-pool approach. Biom J 2024; 66(1):e2200312
- Omori T, Hayashi M. The assessment and communication of genotoxicity test results: moving beyond binary. Mutat Res Genet Toxicol Environ Mutagen 2024;893:503722
- Sano Y, Tanaka S, Sato T. Estimating cure proportion in cancer clinical trials using flexible parametric cure models. BJC Rep 2024;2(1):61
- Tanaka S, Igarashi A, De Moor R, Li N, Hirozane M, Hong LW, Wu DB, Yu DY, Hashim M, Hutton B; EVERSANA Group; Tantakoun K, Samjoo IA, Cameron C. A targeted review of worldwide indirect treatment comparison guidelines and best practices. Value Health 2024:S1098-3015(24)02402-1
- Tanaka S, Muramatsu Y, Inoue K. Clarifying causal effects of interest and underlying assumptions in randomized and nonrandomized clinical trials in oncology using directed acyclic graphs and single world intervention graphs. JCO Clin Cancer Informatics 2024: 8:e2300262
Laboratory
Program-specific Professor:Shiro Tanaka
Program-specific Professor:Takashi Omori
Program-spesific Assistant Professor:Masatomo Omiya
Program-specific Researcher:Yumi Takagi
TEL:+81-75-753-9322
E-mail:kyotocbc-office(at)umin.ac.jp
URL:http://www.cbc.med.kyoto-u.ac.jp
